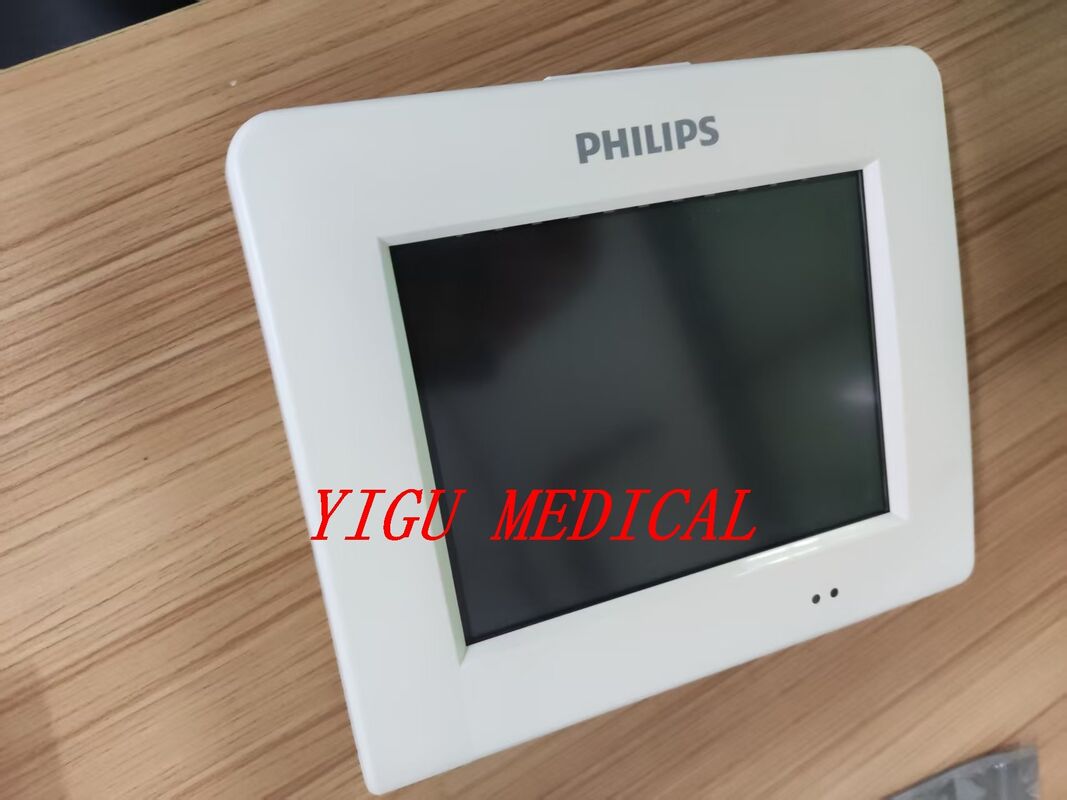
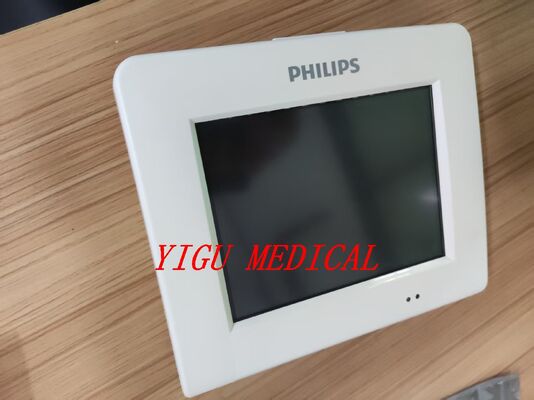
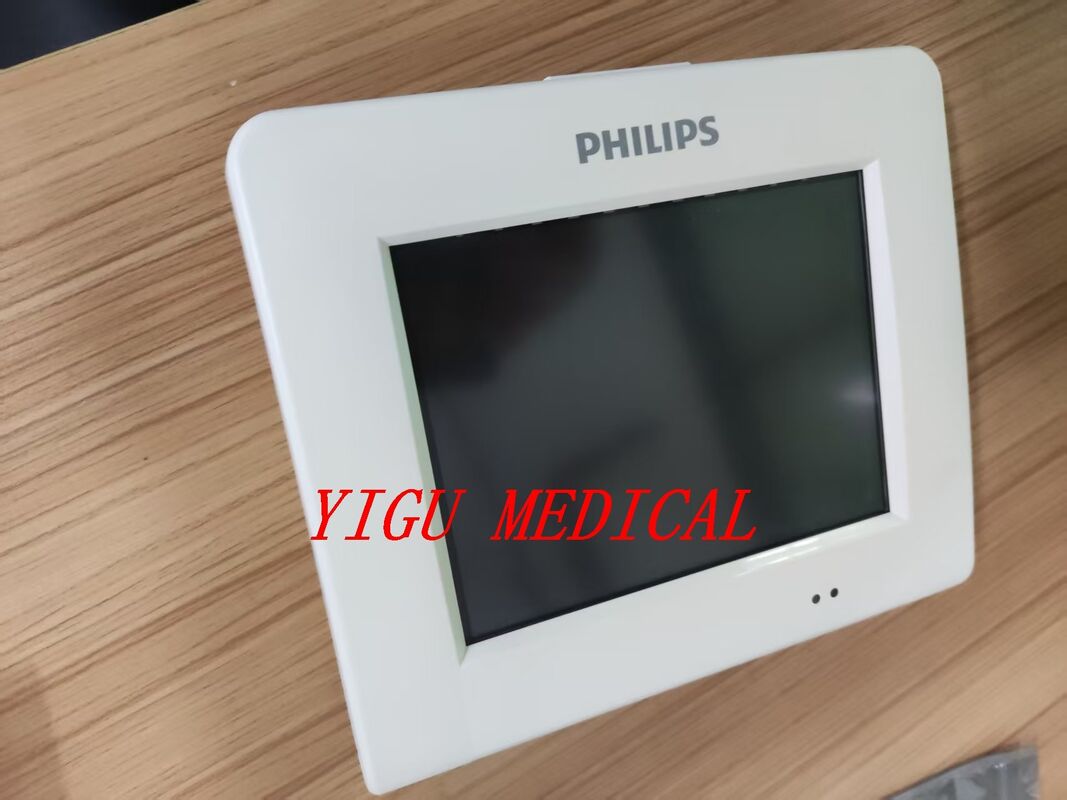
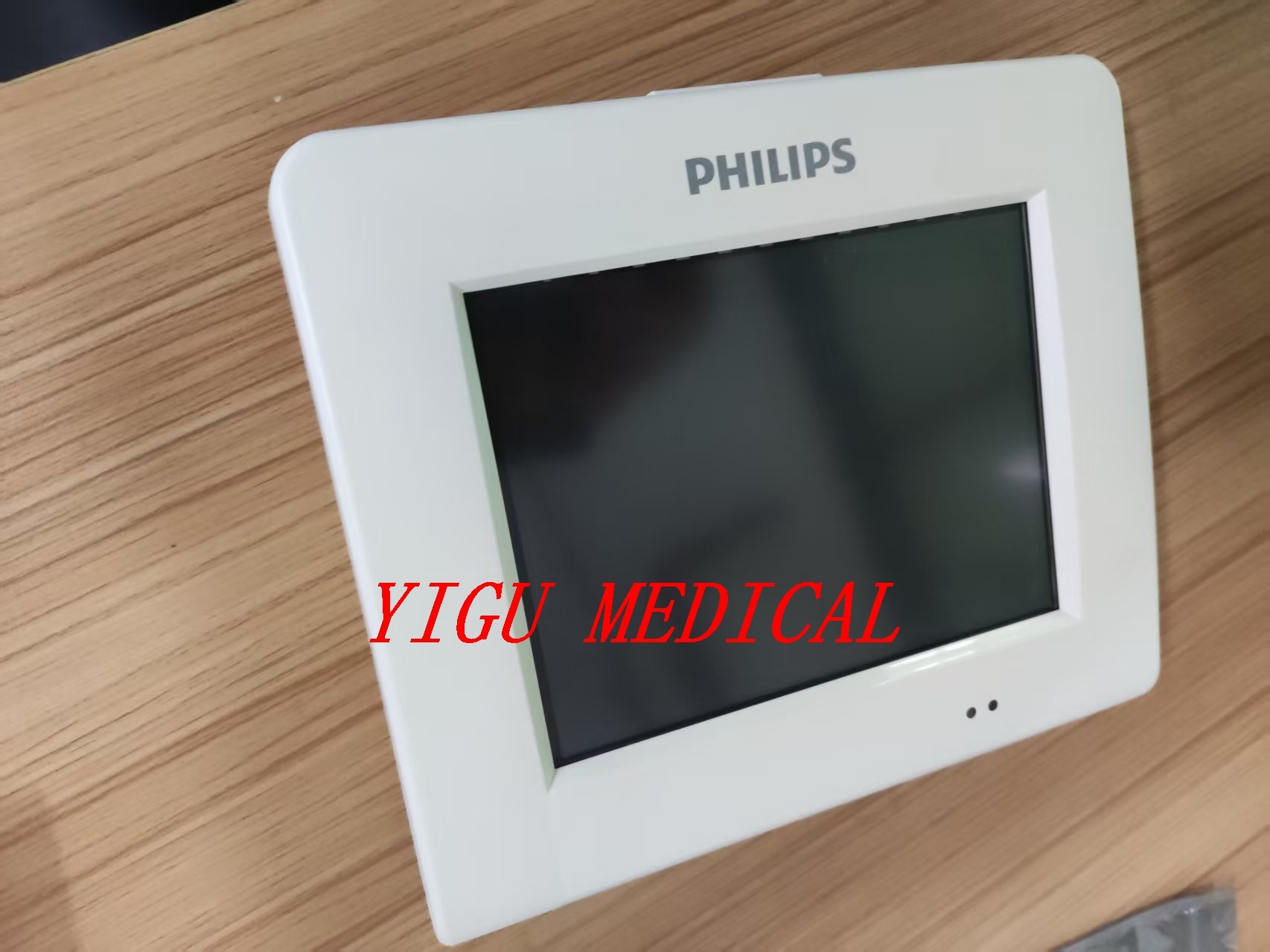
메시지를 남겨주세요
곧 다시 연락 드리겠습니다!
 귀하의 메시지는 20-3,000 자 사이 여야합니다!
귀하의 메시지는 20-3,000 자 사이 여야합니다!
 이메일을 확인하십시오!
이메일을 확인하십시오!
제출
정보가 많을수록 커뮤니케이션이 향상됩니다.
이 씨
- 이 씨
- 부인
승인
성공적으로 제출되었습니다!
곧 다시 연락 드리겠습니다!
승인
메시지를 남겨주세요
곧 다시 연락 드리겠습니다!
 귀하의 메시지는 20-3,000 자 사이 여야합니다!
귀하의 메시지는 20-3,000 자 사이 여야합니다!
 이메일을 확인하십시오!
이메일을 확인하십시오!
제출